Netherlands Neurology Clinical Trials Market Analysis
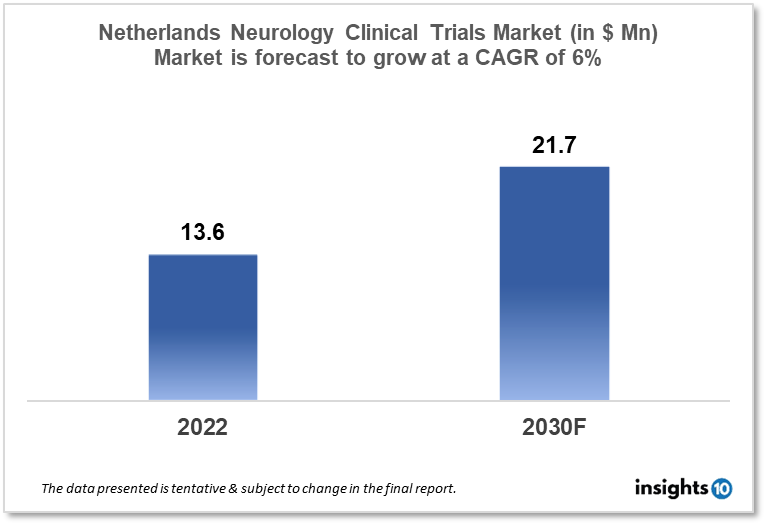
Netherlands's Neurology Clinical Trials market is projected to grow from $13.6 Mn in 2022 to $21.7 Mn by 2030, registering a CAGR of 6% during the forecast period of 2022-30. The market will be driven by the modern healthcare infrastructure and an innovative & collaborative research environment. The market is segmented by phase, by study design & by indication. Some of the major players include Biogen Netherlands BV, Roche Holding AG & Johnson & Johnson.
Buy Now

Netherlands Neurology Clinical Trials Market Executive Summary
Netherlands's Neurology Clinical Trials market is projected to grow from $13.6 Mn in 2022 to $21.7 Mn by 2030, registering a CAGR of 6% during the forecast period of 2022-30. Alzheimer's and dementia fatalities in the Netherlands reached 18,148 in 2020, accounting for 14.31% of total deaths, based on the most recent WHO statistics. The Netherlands ranks seventh in the world with an age-adjusted death rate of 33.78 per 100,000 inhabitants. There are around 120,000 individuals in the Netherlands who have active epilepsy.
The Netherlands has a reasonably robust neurology clinical trials market owing to the country's well-established healthcare system, research infrastructure, and supportive regulatory environment. The Netherlands has a long history of excelling in medical research, notably in the area of neuroscience, and it is home to many world-class university medical institutes such as Amsterdam University Medical Centers, Radboud University Medical Center & University Medical Center Groningen that perform clinical trials in the subject. Moreover, companies such as Biogen, UCB, Janssen & AbbVie have also been keen on performing clinical trials in the Netherlands.
The nation also has a well-developed clinical research regulatory system, including the Central Council on Research Involving Human Subjects (CCMO), which monitors clinical trials involving human volunteers. The CCMO guarantees that clinical trials in the Netherlands are ethical, transparent, and under national and international legislation.
Market Dynamics
Market Growth Drivers
The Netherlands has been an active participant in neurology clinical trials. The modern healthcare infrastructure, along with an innovative research environment, is one of the major drivers of the country's rise in neurology clinical trials. The country's healthcare system is well-known for providing high-quality treatment and efficient administration. The nation also has a well-developed clinical research regulatory structure, including the Central Council on Research Involving Human Subjects (CCMO), which regulates clinical trials with human subjects.
Moreover, the Netherlands has an extensive network of academic universities and research organizations dedicated to neurological research. This network facilitates cooperation among researchers, physicians, and industry partners, creating an environment favorable to the development of innovative treatments and therapies for neurological illnesses.
Market Restraints
there are a number of constraints that might limit the expansion of neurology clinical trials in the Netherlands. The rising complexity and expense of clinical trials are one of the most critical issues. Clinical trials for neurological illnesses sometimes need a large number of patients and sophisticated research designs, making them costly to undertake. This may restrict the number of studies that may be carried out in the Netherlands, particularly for smaller enterprises or academic institutions with little finance. Another difficulty is growing competition from other nations, notably those in Eastern Europe and Asia, which are drawing more clinical trials due to cheaper prices and less severe regulations. Competitiveness may result in fewer clinical trials being done in the Netherlands, limiting the country's ability to contribute to the discovery of innovative therapies for neurological illnesses.
Competitive Landscape
Key Players
- Biogen Netherlands BV
- Roche Holding AG
- Pfizer Inc.
- Novartis AG
- Janssen Pharmaceutical (Johnson & Johnson)
- UCB Pharma B.V. (NLD)
- AstraZeneca plc
- ICON plc
- CHDR (Centre for Human Drug Research)
Notable Deals
January 2023, The FDA approved Eisai, a Biogen Alzheimer's drug.
Healthcare Policies and Regulatory Landscape
The Netherlands has completely adopted the EU Clinical Trials Directive (CTD). An identical Clinical Trial Application (CTA) document must be submitted in tandem with an authorized Ethics Committee and the Centrale Commissie Mensgebonden Onderzoek (CCMO=Competent Authority) before the start of a clinical study. The research may begin after the Ethics Committee has given its approval and the Competent Authority has issued a declaration of no objection.
1. Executive Summary
1.1 Disease Overview
1.2 Global Scenario
1.3 Country Overview
1.4 Healthcare Scenario in Country
1.5 Clinical Trials Regulation in Country
1.6 Recent Developments in the Country
2. Market Size and Forecasting
2.1 Market Size (With Excel and Methodology)
2.2 Market Segmentation (Check all Segments in Segmentation Section)
3. Market Dynamics
3.1 Market Drivers
3.2 Market Restraints
4. Competitive Landscape
4.1 Major Market Share
4.2 Key Company Profile (Check all Companies in the Summary Section)
4.2.1 Company
4.2.1.1 Overview
4.2.1.2 Product Applications and Services
4.2.1.3 Recent Developments
4.2.1.4 Partnerships Ecosystem
4.2.1.5 Financials (Based on Availability)
5. Reimbursement Scenario
5.1 Reimbursement Regulation
6. Methodology and Scope
Neurology Clinical Trials Market Segmentation
By Phase (Revenue, USD Billion):
- Phase I
- Phase II
- Phase III
- Phase IV
By Study Design Outlook (Revenue, USD Billion):
- Epilepsy
- Parkinson's Disease (PD)
- Huntington's Disease
- Stroke
- Traumatic Brain Injury (TBI)
- Amyotrophic Lateral Sclerosis (ALS)
- Muscle regeneration
- Others
By Indication Outlook (Revenue, USD Billion):
- Interventional
- Observational
- Expanded Access
Methodology for Database Creation
Our database offers a comprehensive list of healthcare centers, meticulously curated to provide detailed information on a wide range of specialties and services. It includes top-tier hospitals, clinics, and diagnostic facilities across 30 countries and 24 specialties, ensuring users can find the healthcare services they need.
Additionally, we provide a comprehensive list of Key Opinion Leaders (KOLs) based on your requirements. Our curated list captures various crucial aspects of the KOLs, offering more than just general information. Whether you're looking to boost brand awareness, drive engagement, or launch a new product, our extensive list of KOLs ensures you have the right experts by your side. Covering 30 countries and 36 specialties, our database guarantees access to the best KOLs in the healthcare industry, supporting strategic decisions and enhancing your initiatives.
How Do We Get It?
Our database is created and maintained through a combination of secondary and primary research methodologies.
1. Secondary Research
With many years of experience in the healthcare field, we have our own rich proprietary data from various past projects. This historical data serves as the foundation for our database. Our continuous process of gathering data involves:
- Analyzing historical proprietary data collected from multiple projects.
- Regularly updating our existing data sets with new findings and trends.
- Ensuring data consistency and accuracy through rigorous validation processes.
With extensive experience in the field, we have developed a proprietary GenAI-based technology that is uniquely tailored to our organization. This advanced technology enables us to scan a wide array of relevant information sources across the internet. Our data-gathering process includes:
- Searching through academic conferences, published research, citations, and social media platforms
- Collecting and compiling diverse data to build a comprehensive and detailed database
- Continuously updating our database with new information to ensure its relevance and accuracy
2. Primary Research
To complement and validate our secondary data, we engage in primary research through local tie-ups and partnerships. This process involves:
- Collaborating with local healthcare providers, hospitals, and clinics to gather real-time data.
- Conducting surveys, interviews, and field studies to collect fresh data directly from the source.
- Continuously refreshing our database to ensure that the information remains current and reliable.
- Validating secondary data through cross-referencing with primary data to ensure accuracy and relevance.
Combining Secondary and Primary Research
By integrating both secondary and primary research methodologies, we ensure that our database is comprehensive, accurate, and up-to-date. The combined process involves:
- Merging historical data from secondary research with real-time data from primary research.
- Conducting thorough data validation and cleansing to remove inconsistencies and errors.
- Organizing data into a structured format that is easily accessible and usable for various applications.
- Continuously monitoring and updating the database to reflect the latest developments and trends in the healthcare field.
Through this meticulous process, we create a final database tailored to each region and domain within the healthcare industry. This approach ensures that our clients receive reliable and relevant data, empowering them to make informed decisions and drive innovation in their respective fields.
To request a free sample copy of this report, please complete the form below.
We value your inquiry and offer free customization with every report to fulfil your exact research needs.










































