Brazil Cardiac Arrhythmia Therapeutics Market
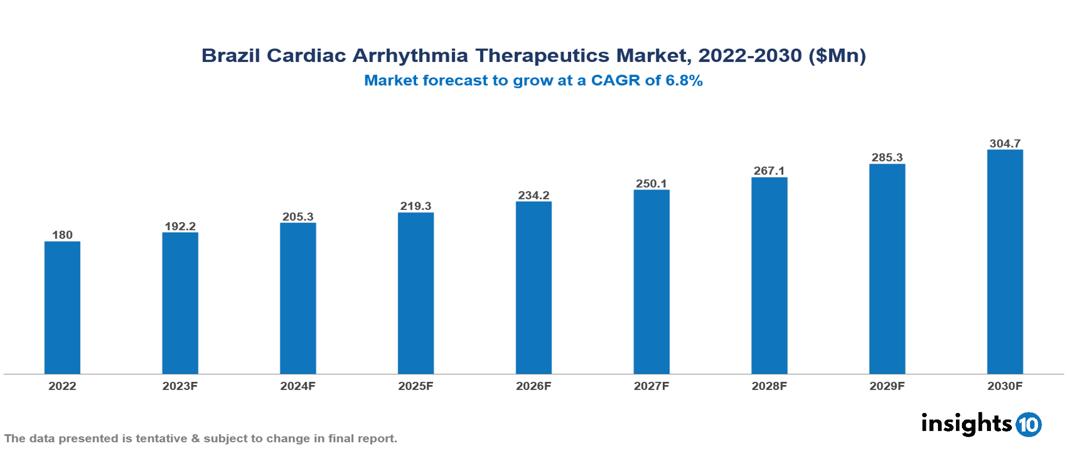
Brazil Cardiac Arrhythmia Therapeutics Market valued at $180 Mn in 2022, projected to reach $305 Mn by 2030 with a 6.8% CAGR. The key drivers of this industry include the rising burden of cardiac conditions, technological advancements, and growing healthcare expenditure. The industry is primarily dominated by players such as Abbott, Medtronic, Biotronik, Biolab Sanus, Johnson & Johnson, and Boston Scientific among others.
Buy Now

Brazil Cardiac Arrhythmia Therapeutics Market Analysis Executive Summary
Brazil Cardiac Arrhythmia Therapeutics Market valued at $180 Mn in 2022, projected to reach $305 Mn by 2030 with a 6.8% CAGR.
Cardiac arrhythmia is characterized by an irregular heartbeat, presenting as either a rapid, slow, or irregular pattern of heartbeats. This condition can disrupt the regular functioning of the heart, impacting its efficiency in pumping blood. Common factors contributing to cardiac arrhythmias encompass underlying heart conditions like coronary artery disease, heart valve disorders, high blood pressure, diabetes, smoking, alcohol consumption, and stress. Symptoms may include palpitations, chest discomfort, dizziness, fainting, and fatigue, typically diagnosed through ECG or EKG tests. Treatment options include various approaches such as anti-arrhythmic drugs, beta-blockers, and calcium channel blockers, all aimed at regulating heart rhythm and rate. Additionally, implantable devices like pacemakers or defibrillators are employed. Renowned companies in the field, such as Medtronic, Boston Scientific, Abbott Laboratories, and Johnson & Johnson, actively contribute to advancing innovative medical devices and pharmaceuticals for cardiac arrhythmia treatments.
The approximate prevalence of atrial fibrillation is about 2.5% in Brazil. The market is being driven by crucial factors such as the rising burden of cardiovascular diseases, technological advancements in the therapeutics industry, and growing healthcare expenditure. However, conditions such as limited affordability, health system challenges, and lack of local manufacturing limit the growth and potential of the market.
Market Dynamics
Market Growth Drivers
Increase in prevalence of CVDs: Brazil possesses a sizable and aging population, factors that pose significant risks for the emergence of cardiac arrhythmias. The elderly demographic in Brazil is anticipated to reach 43 Mn by 2030, indicating a considerable and expanding demographic for arrhythmia therapeutics. The estimated prevalence of atrial fibrillation is about 2.5% in Brazil. The escalating trend of urbanization, coupled with unhealthy dietary practices and insufficient physical activity, contributes to the rising incidence of CVDs which further propels the growth of the market.
Improved healthcare expenditure: The Brazilian government has instituted diverse measures to enhance healthcare accessibility, including the implementation of the Unified Health System (SUS) and the National Cardiovascular Health Plan. Simultaneously, the private healthcare sector in Brazil is undergoing substantial expansion, prompting greater investments in cutting-edge medical technologies and advanced treatments for arrhythmias, such as ablation catheters and pacemakers. The overall healthcare expenditure in Brazil is anticipated to increase in the foreseeable future, creating a more favorable climate for investments in innovative therapeutics for arrhythmia.
Technological advancements: The increasing acceptance of minimally invasive techniques such as catheter ablation in the treatment of arrhythmias is favored for its shorter recovery periods and reduced likelihood of complications. This preference contributes to the expanding market for specialized devices and technologies. Concurrently, pharmaceutical firms are proactively creating novel and enhanced medications for arrhythmia management, incorporating personalized medicine strategies tailored to the specific needs of individual patients. This ongoing development serves as an additional catalyst for the growth of the market.
Market Restraints
Limited affordability: Despite having private insurance coverage, individuals frequently encounter elevated out-of-pocket costs associated with arrhythmia treatments, encompassing medications, devices, and procedures. This financial burden serves as a significant obstacle, particularly for those with lower incomes. The intricacies involved in the reimbursement procedures for arrhythmia treatments, both from public and private insurers, can lead to delays and uncertainties, exacerbating difficulties in accessing and affording necessary care.
Health system challenges: Brazil faces a scarcity of cardiologists and electrophysiologists (EPs) who specialize in diagnosing and treating arrhythmias. This shortage contributes to extended waiting periods for consultations and procedures, adversely affecting patient care and impeding market growth. The existing specialists are predominantly clustered in major cities, resulting in rural areas having restricted access to high-quality care. This imbalance in healthcare distribution creates disparities in treatment choices and outcomes, ultimately impacting the overall potential of the market.
Limited local manufacturing: Brazil heavily depends on imported treatments for arrhythmia, resulting in increased expenses attributable to tariffs and additional import-related fees. The constrained domestic manufacturing capacity also poses a limitation on the potential growth of the market.
Healthcare Policies and Regulatory Landscape
In Brazil, the regulatory authority responsible for overseeing therapeutics, including pharmaceuticals, medical devices, and biologics, is the Agência Nacional de Vigilância Sanitária (ANVISA). ANVISA is the National Health Surveillance Agency and operates under the Ministry of Health. The agency plays a crucial role in ensuring the safety, efficacy, and quality of therapeutic products in the Brazilian market. To obtain licensure for a therapeutic product in Brazil, manufacturers must submit a comprehensive dossier containing detailed information on the product's formulation, manufacturing processes, preclinical and clinical data, and risk management plans.
The environment for new entrants in the Brazilian therapeutics market involves navigating a complex regulatory landscape. While ANVISA aims to ensure public health and safety, the regulatory processes can be rigorous, requiring significant documentation and adherence to specific standards. The time and resources needed for regulatory approval can pose challenges for new entrants, and thorough preparation and engagement with regulatory authorities are crucial for success. Overall, despite the challenges, the Brazilian market presents opportunities for innovative therapeutics, and successful entry requires a strategic approach to regulatory compliance and market dynamics.
Competitive Landscape
Key Players
- Abbott
- Boston Scientific Corporation
- Medtronic
- Biotronik
- Johnson & Johnson
- Siemens Healthineers
- Stryker Corporation
- Biolab Sanus
- Bayer
- Merck
1. Executive Summary
1.1 Disease Overview
1.2 Global Scenario
1.3 Country Overview
1.4 Healthcare Scenario in Country
1.5 Patient Journey
1.6 Health Insurance Coverage in Country
1.7 Active Pharmaceutical Ingredient (API)
1.8 Recent Developments in the Country
2. Market Size and Forecasting
2.1 Epidemiology of Disease
2.2 Market Size (With Excel & Methodology)
2.3 Market Segmentation (Check all Segments in Segmentation Section)
3. Market Dynamics
3.1 Market Drivers
3.2 Market Restraints
4. Competitive Landscape
4.1 Major Market Share
4.2 Key Company Profile (Check all Companies in the Summary Section)
4.2.1 Company
4.2.1.1 Overview
4.2.1.2 Product Applications and Services
4.2.1.3 Recent Developments
4.2.1.4 Partnerships Ecosystem
4.2.1.5 Financials (Based on Availability)
5. Reimbursement Scenario
5.1 Reimbursement Regulation
5.2 Reimbursement Process for Diagnosis
5.3 Reimbursement Process for Treatment
6. Methodology and Scope
Brazil Cardiac Arrhythmia Therapeutics Market Segmentation
By Test Equipment
- Electrocardiogram (ECG)
- Holter monitor
- Others
By Site of Origin
- Atrial Fibrillation
- Sinus Bradycardia
- Atrial Tachycardia
- Atrial Flutter
- Premature Atrial Contractions (PACS)
- Others
By Type
- Supraventricular Tachycardias
- Ventricular Arrhythmias
- Bradyarrhythmia’s
By Drug Type
- Antiarrhythmic drugs
- Calcium channel blockers
- Beta-blockers
- Anticoagulants
- Others
By Mode of Administration
- Injectable
- Oral
- Others
By Distribution channel
- Hospital pharmacies
- Retail pharmacies
- Online pharmacies
Methodology for Database Creation
Our database offers a comprehensive list of healthcare centers, meticulously curated to provide detailed information on a wide range of specialties and services. It includes top-tier hospitals, clinics, and diagnostic facilities across 30 countries and 24 specialties, ensuring users can find the healthcare services they need.
Additionally, we provide a comprehensive list of Key Opinion Leaders (KOLs) based on your requirements. Our curated list captures various crucial aspects of the KOLs, offering more than just general information. Whether you're looking to boost brand awareness, drive engagement, or launch a new product, our extensive list of KOLs ensures you have the right experts by your side. Covering 30 countries and 36 specialties, our database guarantees access to the best KOLs in the healthcare industry, supporting strategic decisions and enhancing your initiatives.
How Do We Get It?
Our database is created and maintained through a combination of secondary and primary research methodologies.
1. Secondary Research
With many years of experience in the healthcare field, we have our own rich proprietary data from various past projects. This historical data serves as the foundation for our database. Our continuous process of gathering data involves:
- Analyzing historical proprietary data collected from multiple projects.
- Regularly updating our existing data sets with new findings and trends.
- Ensuring data consistency and accuracy through rigorous validation processes.
With extensive experience in the field, we have developed a proprietary GenAI-based technology that is uniquely tailored to our organization. This advanced technology enables us to scan a wide array of relevant information sources across the internet. Our data-gathering process includes:
- Searching through academic conferences, published research, citations, and social media platforms
- Collecting and compiling diverse data to build a comprehensive and detailed database
- Continuously updating our database with new information to ensure its relevance and accuracy
2. Primary Research
To complement and validate our secondary data, we engage in primary research through local tie-ups and partnerships. This process involves:
- Collaborating with local healthcare providers, hospitals, and clinics to gather real-time data.
- Conducting surveys, interviews, and field studies to collect fresh data directly from the source.
- Continuously refreshing our database to ensure that the information remains current and reliable.
- Validating secondary data through cross-referencing with primary data to ensure accuracy and relevance.
Combining Secondary and Primary Research
By integrating both secondary and primary research methodologies, we ensure that our database is comprehensive, accurate, and up-to-date. The combined process involves:
- Merging historical data from secondary research with real-time data from primary research.
- Conducting thorough data validation and cleansing to remove inconsistencies and errors.
- Organizing data into a structured format that is easily accessible and usable for various applications.
- Continuously monitoring and updating the database to reflect the latest developments and trends in the healthcare field.
Through this meticulous process, we create a final database tailored to each region and domain within the healthcare industry. This approach ensures that our clients receive reliable and relevant data, empowering them to make informed decisions and drive innovation in their respective fields.
To request a free sample copy of this report, please complete the form below.
We value your inquiry and offer free customization with every report to fulfil your exact research needs.